Atomic and Molecular Physics Homework 4
Chasse_neige
Question1
A helium atom is excited from the ground state to the autoionizing state
The Hamiltonian is
where
So the Schrödinger equation gives that the zero-order wavefunction takes the form of
So we get the zero-order energy
For the first-order perturbation of the energy, consider the perturbation of the Hamiltonian
Let
and we'll get
Where the parameters is the direct integral
and the exchange integral
Given that the factor
Using the specific form of the radial wavefunction
Because these integrals are nearly impossible to be calculated by hand, so I'll use mathematica to give a numerical answer:
ClearAll["Global`*"]
R[Z_, n_, l_, r_] := Module[{rho, Nnorm}, rho = (2 Z r)/n;
Nnorm = Sqrt[(2 Z/n)^3*(n - l - 1)!/(2 n*(n + l)!)];
Nnorm*Exp[-rho/2]*rho^l*LaguerreL[n - l - 1, 2 l + 1, rho]];
SlaterIntegral[k_, Z1_, n1_, l1_, Z2_, n2_, l2_, Z3_, n3_, l3_, Z4_,
n4_, l4_] :=
Module[{integrand, r1, r2, radialPart1, radialPart2, result},
result =
Integrate[
r1^2*R[Z1, n1, l1, r1]*
R[Z3, n3, l3,
r1]*(Integrate[
r2^2*R[Z2, n2, l2, r2]*
R[Z4, n4, l4, r2]*(r2^k/r1^(k + 1)), {r2, 0, r1}] +
Integrate[
r2^2*R[Z2, n2, l2, r2]*
R[Z4, n4, l4, r2]*(r1^k/r2^(k + 1)), {r2, r1,
Infinity}]), {r1, 0, Infinity}, Assumptions -> {r1 > 0}];
Return[result];];
Z2s = 2; n2s = 2; l2s = 0;
Z4p = 1; n4p = 4; l4p = 1;
J = SlaterIntegral[0, Z2s, n2s, l2s, Z4p, n4p, l4p, Z2s, n2s, l2s,
Z4p, n4p, l4p];
K = SlaterIntegral[1, Z2s, n2s, l2s, Z4p, n4p, l4p, Z4p, n4p, l4p,
Z2s, n2s, l2s];
Print[J];
Print[K];The script gives out that
and
So we can get that the first-order energy correction
So the energy for the this autoionizing level at singlet and triplet states are
For the ground state where the two electrons are at the
while the first-order correction gives out that
So the energy for the ground state is
So the energy gap between the ground state and this exited state is
So the wavelength of the UV radiation are
(b) Find the velocity of the electron emitted in the autoionizing process in which the autoionizing level
For the
So the total energy of the emitted electron is
Therefore, the velocity of the emitted electron is
Question2
Consider the ground state of zirconium which has a ground state’s term symbol of
Zirconium has atomic number
The remaining
Thus the complete ground‑state electron configuration of
(b) According to the given term symbol, what are the spin angular momentum
For a term symbol
- Spin multiplicity
. - Orbital symbol
. - Total angular momentum
.
(c) Determine all possible values of
The valence electrons that determine the term are the two 4d electrons (the closed shells and the filled 5s subshell contribute
Hund’s first rule states that for a given electron configuration, the term with the highest multiplicity (largest
(d) Determine all possible values of
Each
However, or equivalent electrons (same
(e) Determine all possible values of the total angular momentum
For a given
With
Hund’s third rule states:
- For a subshell less than half filled, the level with the smallest
is lowest. - For a subshell more than half filled, the level with the largest
is lowest.
The
Question3
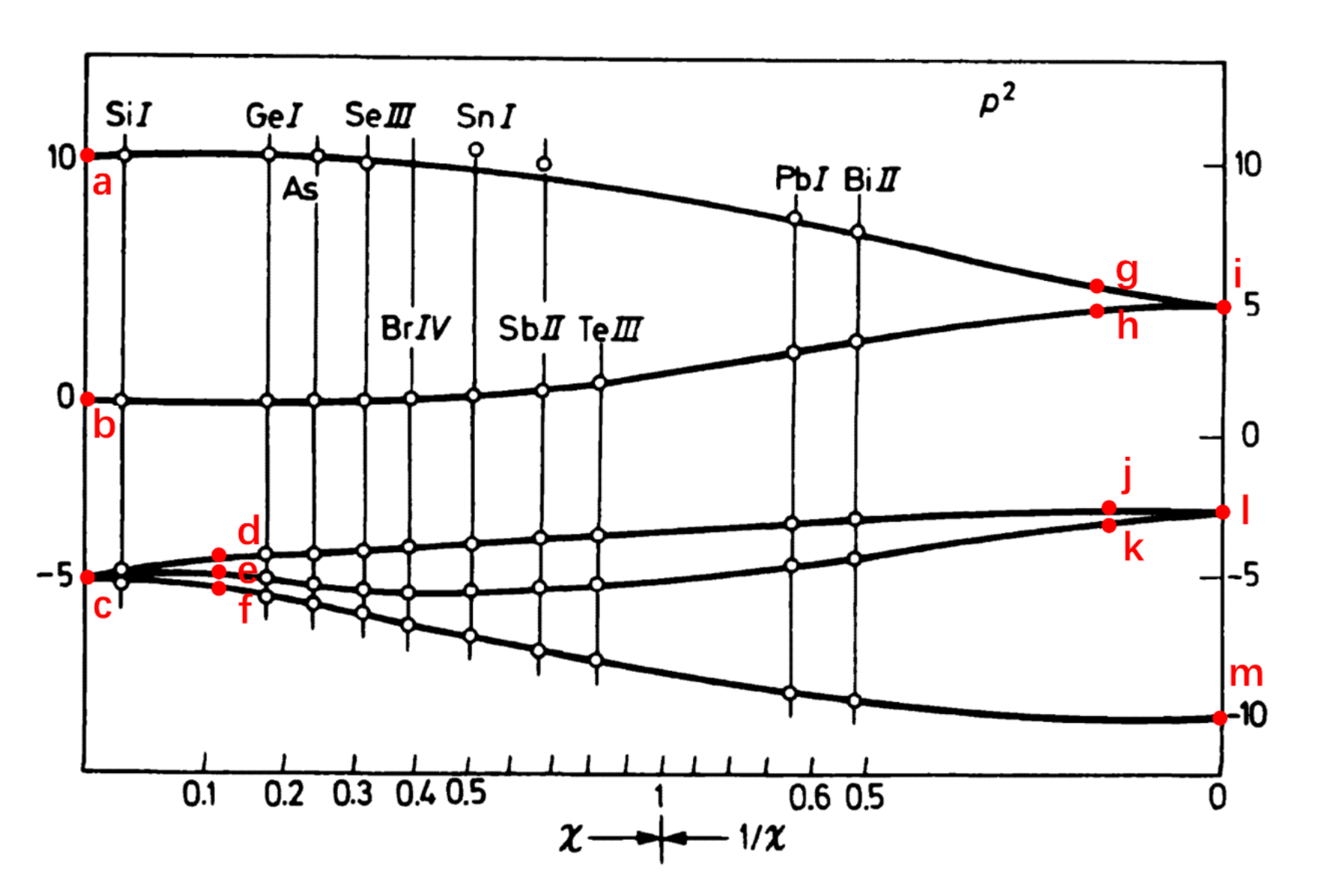
(a) For atoms or ions whose two valence electrons are in the same
For left side of the picture, the atomic number is quite low, so it's proper to describe these states using the effect of
According to the Hund's Rules, the ordering of the states are:
is the ground term (lowest energy). Highest Orbital (
) for same : is the next lowest. is the highest energy term.
Considering the Fine Structure Spin-Orbit Splitting:
The
term ( ) splits into . For a less-than-half-filled shell, the lowest has the lowest energy. . The
term ( ) has only : . The
term ( ) has only : .
So the energy order at low
: Both electrons in . Lowest energy (spin-orbit favors lower ). Allowed : 0. : One in , one in . Intermediate energy. Allowed : 1, 2. : Both electrons in . Highest energy. Allowed : 0, 2.
States with the same total angular momentum
: Lowest ( ) Lowest ( ) . Highest ( ) Highest ( ) . : Only one state ( ) Middle group ( ) . : Lower ( ) Middle group ( ) . Higher ( ) Highest group ( ) .
According to the analysis above, we can get the assigned term symbols for the points given:
Left Side (L-S Coupling Region):
(a)
(b)
(c)
The bottom cluster originates from the
(f)
(d)
(e)
Right Side (j-j Coupling Region): In
(i)
(l)
(m)
(g)
(h)
(j)
(k)
